 The representation of a molecular or ionic structure by several structures is called resonance Each dot represents one valence electron, and the fact that they are placed between the two atoms means that they are being shared bas a covalent bond. When several dot structures are reasonable for a molecule or ion, they all contribute to the molecular or ionic structure making it more stable. Lewis dot structures are useful in explaining the chemical bonding in molecules or ions. 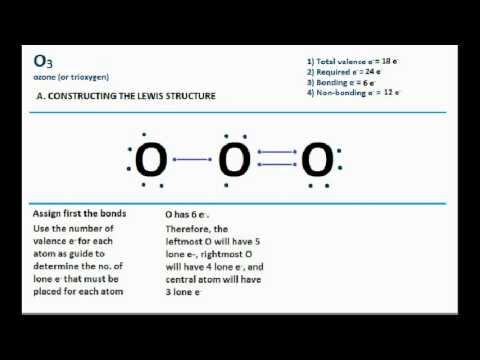
Lewis, who introduced it in his 1916 article The Atom and the. The Lewis structure was named after Gilbert N. A Lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds. Lewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures, are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs of electrons that may exist in the molecule. 4 Published in the journal Nature, the study, Structural and Spectroscopic Characterization of an Einsteinium Complex, was co-led by Berkeley Lab scientist Rebecca Abergel and Los Alamos National. Einsteinium was discovered by Albert Ghiorso in 1952. The boiling point of Einsteinium is 996 C & the melting point of Einsteinium is 860 C. 
Home Einsteinium lewis dot structure Copy of Nickelman Element SuperheroesĮlement Superheroes Bohr model for Einsteinium: Electron Configuration: Rn 5f11 7s2 Lewis Dot Structure 10 facts about the Einsteinium 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
